
676 Chapter 20
Copyright © 2017 Pearson Education, Inc.
Solutions to Problems
1.
d-Ribose is an aldopentose.
d-Sedoheptulose is a ketoheptose.
d-Mannose is an aldohexose.
2.
Notice that an l-sugar is the mirror image of a d-sugar.
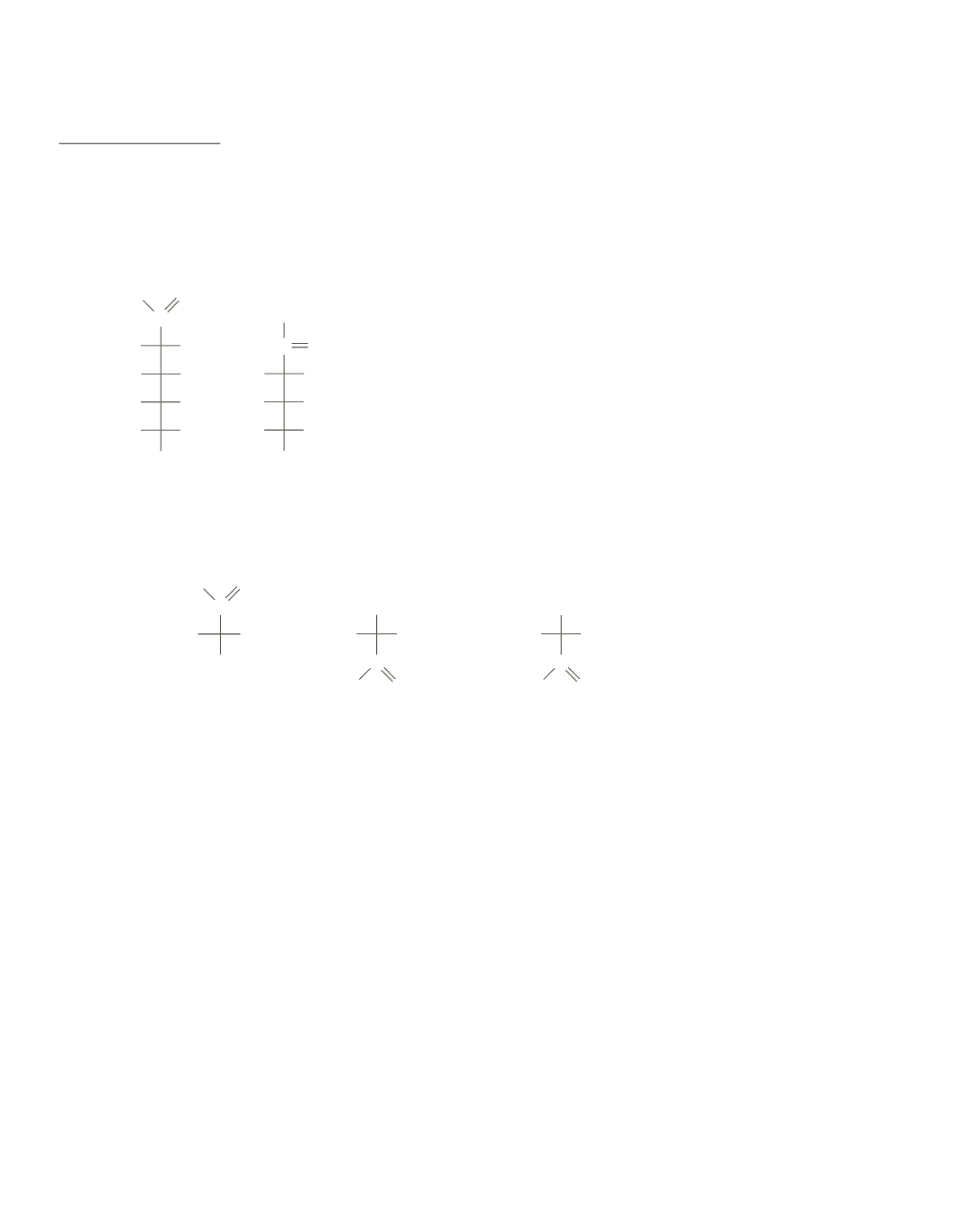
C
CH
2
OH
H HO
OH
H
H HO
H HO
O H
L
-glucose
CH
2
OH
C
CH
2
OH
OH H
H HO
H HO
O
L
-fructose
3.
First determine whether the structure represents
R
-glyceraldehyde or
S
-glyceraldehyde. Then, because
R
-glyc-
eraldehyde
=
d-glyceraldehyde and
S
-glyceraldehyde
=
l-glyceraldehyde, you can answer the question.
C
H
OH
HOCH
a.
b.
c.
2
H
CH
2
OH
HO
O H
CH
2
OH
H HO
L
-glyceraldehyde
L
-glyceraldehyde
D
-glyceraldehyde
H O
C
O H
C
4.
a.
enantiomers because they are mirror images
b.
diastereomers because the configuration of one asymmetric center is the same in both and the configu-
ration of the other asymmetric center is the opposite in both
5.
a.
d-ribose
b.
l-talose
c.
l-allose
d.
l-ribose
6.
a.
d-glucose
=
1
2R,3S,4R,5R
2
-2,3,4,5,6-pentahydroxyhexanal
b.
Because d-mannose is the C-2 epimer of d-glucose, the systematic name of d-mannose can be obtained
just by changing the configuration of the C-2 carbon in the systematic name of d-glucose.
d
@
mannose
=
1
2
S
,3
S
,4
R
,5
R
2
@
2,3,4,5,6
@
pentahydroxyhexanal
c.
d-Galactose is the C-4 epimer of d-glucose. Therefore, each of its carbon atoms, except C-4, has the
same configuration as it has in d-glucose.
d
@
galactose
=
1
2
R
,3
S
,4
S
,5
R
2
@
2,3,4,5,6
@
pentahydroxyhexanal
d.
l-Glucose is the mirror image of d-glucose, so each carbon in l-glucose has the opposite configuration
to that in d-glucose.
l
@
glucose
=
1
2
S
,3
R
,4
S
,5
S
2
@
2,3,4,5,6
@
pentahydroxyhexanal
















