
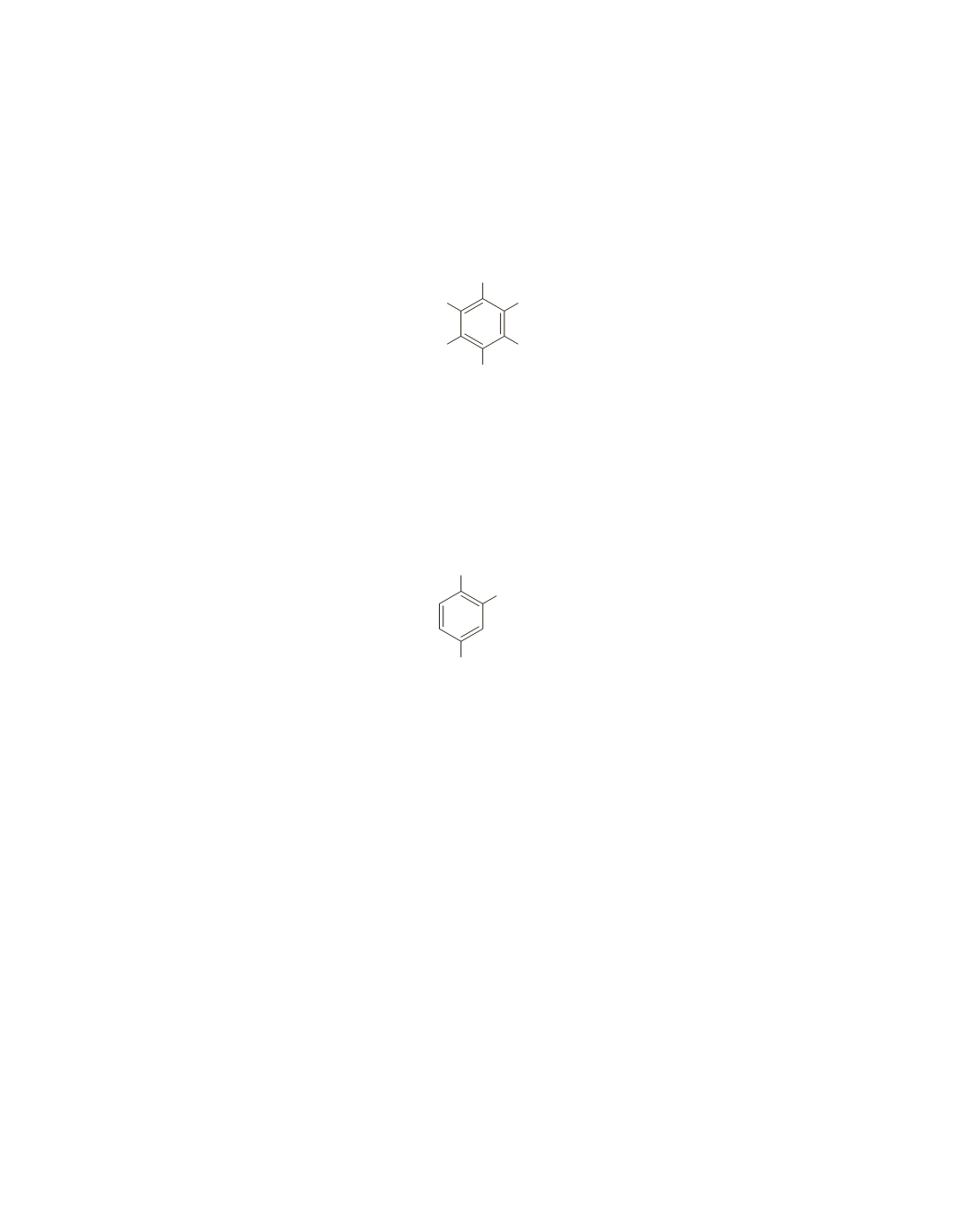
50
Spectroscopy Problems
Copyright © 2017 Pearson Education, Inc.
The substitution pattern for the trisubstituted benzene can be determined from the splitting patterns.
Because the signal (6.5 ppm) that integrates to 1 proton is a doublet, we know that the proton that gives this
signal has only one neighboring proton. Looking at the magnification of the signal at 7.1 ppm, we see that
it is actually two separate signals. One is a singlet; therefore, it is attached to a carbon that is separated by
substituents from the carbons that are attached to protons. The other signal is a doublet that integrates to
1 proton; because it gives a doublet, we know that it is next to the proton that gives the doublet at 6.5 ppm.
X
H
d
H
e
Z
H
c
Y
To determine the relative positions of the substituents, the chemical shifts must be analyzed. Bromine is
the most electronegative substituent and, therefore, must be adjacent to the two protons that give signals
at 7.1 ppm. Thus, Z is Br. The amino group donates its lone-pair electrons into the ring, so it shields
benzene-ring protons. Thus, the signal at 6.5 ppm is from a proton in close proximity to the amino group.
Therefore, X must be the amino group.
The compound that gives the spectrum is shown here.
NH
2
CH
3
Br
18.
The two compounds that produce the spectra have the following structures.
ClCH
2
CH
2
CH
2
Br
1-bromo-3-chloropropane
ClCH
2
CH
2
CH
2
I
1-chloro-3-iodopropane
The number of signals (three) and the splitting patterns are identical for each compound. The only
difference is variations in the chemical shift due to the different electronegativities of bromine and iodine.
Because chlorine is more electronegative than bromine or iodine, the protons bonded to the carbon that is
attached to chlorine has the most deshielded signal (that is, the signal that occurs at the highest frequency).
This is the triplet that occurs at 3.7 ppm in both spectra.
The spectra differ in the signal that occurs at 3.4 ppm in the top spectrum and the signal that appears at
3.6 ppm in the second spectrum. Because bromine is more electronegative than iodine, the protons bonded
to the carbon that is attached to bromine occurs at a higher frequency than the protons bonded to the carbon
that is attached to iodine.
Thus,
1-chloro-3-iodopropane
gives the top spectrum, and
1-bromo-3-chloropropane
gives the bottom
spectrum.
















