
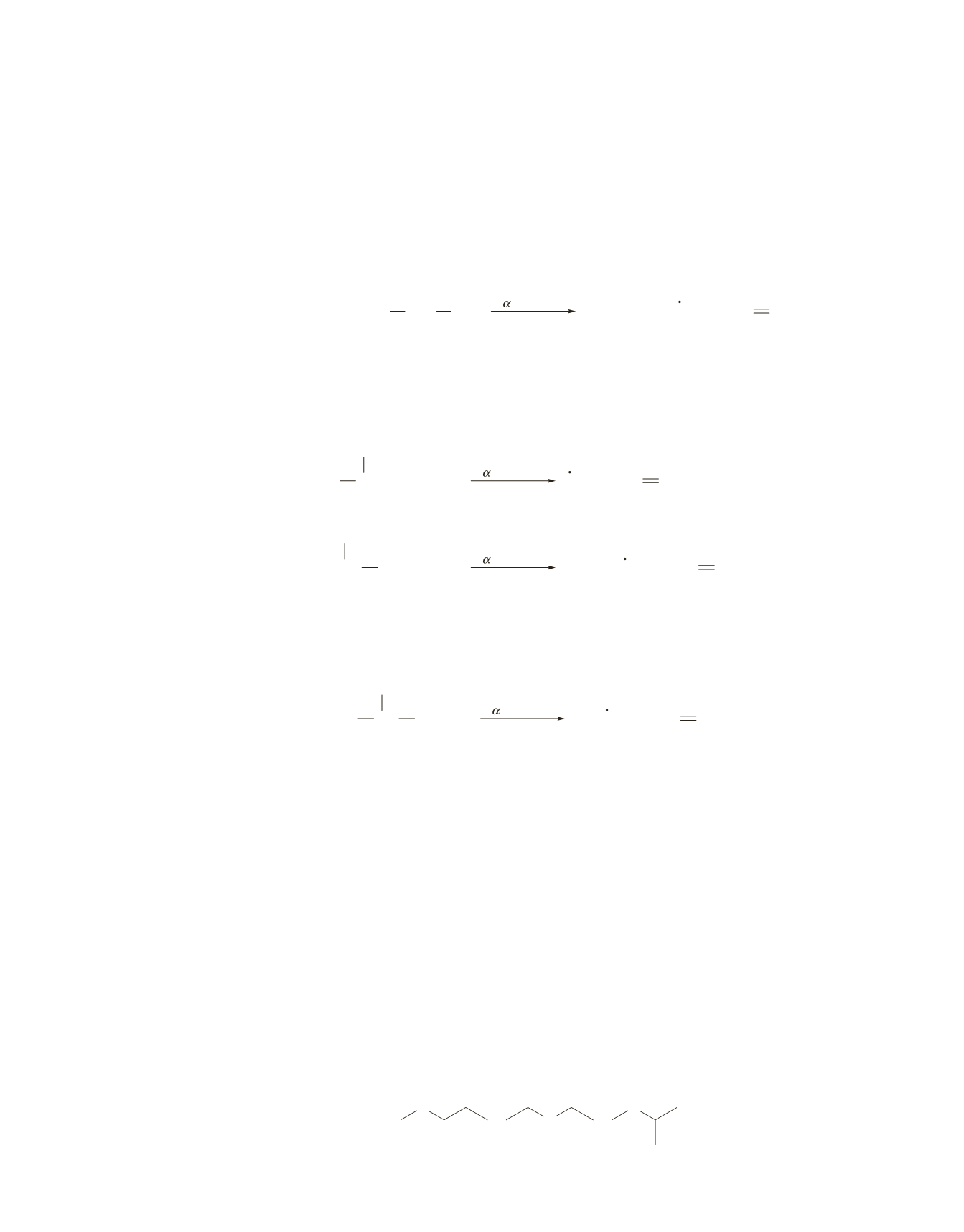
Spectroscopy Problems
41
Copyright © 2017 Pearson Education, Inc.
Answers to Spectroscopy Problems
1.
There are three straight-chain pentanols: 1-pentanol, 2-pentanol, and 3-pentanol. Because the most stable
fragment for an alcohol is the one formed by
a
-cleavage, we can see which of the alcohols forms the base
peak shown in the spectrum (that is, a base peak with
m
>
z
=
45
) as a result of
a
-cleavage.
For 1-pentanol, only one
a
-cleavage is possible. It forms a cationic fragment with
m
>
z
=
31.
CH
2
CH
3
CH
2
CH
2
CH
2
CH
3
CH
2
CH
2
CH
2
OH
m/z
= 31
-cleavage
+
HO CH
2
+
For 2-pentanol, two
a
-cleavages are possible. One forms a cationic fragment with
m
>
z
=
73
and a methyl
radical. The second forms a cationic fragment with
m
>
z
=
45
and a propyl radical. Because a propyl
radical is more stable than a methyl radical, the base peak is expected to have
m
>
z
=
45.
CH
3
CHCH
2
CH
2
CH
3
CH
3
OH
m/z
= 73
-cleavage
+
HO CHCH
2
CH
2
CH
3
+
CH
3
CH CH
2
CH
2
CH
3
OH
-cleavage
CH
3
CH
2
CH
2
m/z
= 45
+
HO CHCH
3
+
CH
3
CHCH
2
CH
2
CH
3
CH
3
OH
m/z
= 73
-cleavage
+
HO CHCH
2
CH
2
CH
3
+
CH
3
CH CH
2
CH
2
CH
3
OH
-cleavage
CH
3
CH
2
CH
2
m/z
= 45
+
HO CHCH
3
+
For 3-pentanol, only one
a
-cleavage is possible because of the symmetry of the molecule.
a
-Cleavage
forms a cationic fragment with
m
>
z
=
59.
CH CH
2
CH
3
CH
3
CH
2
OH
-cleavage
CH
3
CH
2
m/z
= 59
+
HO CHCH
2
CH
3
+
The base peak of the given mass spectrum has
m
>
z
=
45.
Thus, the mass spectrum is that of
2-pentanol
.
We also see a significant fragment at
m
>
z
=
73,
the
m
>
z
value of the other
a
-cleavage product.
2.
First, we must first identify the molecular ion. The molecular ion, the peak that represents the intact starting
compound, has an
m
>
z
=
74.
Now we can use the rule of 13 to determine the molecular formula.
74
13
=
5 carbons with 9 left over
From the rule of 13, we end up with a molecular formula of
C
5
H
14
.
Because the compound is an ether, we
know that it has one oxygen, so we must add one O and subtract one C and four Hs from the molecular
formula. The resulting molecular formula is:
C
4
H
10
O
Three ethers have this molecular formula: methyl propyl ether, diethyl ether, and isopropyl methyl ether.
O
O
O
















