
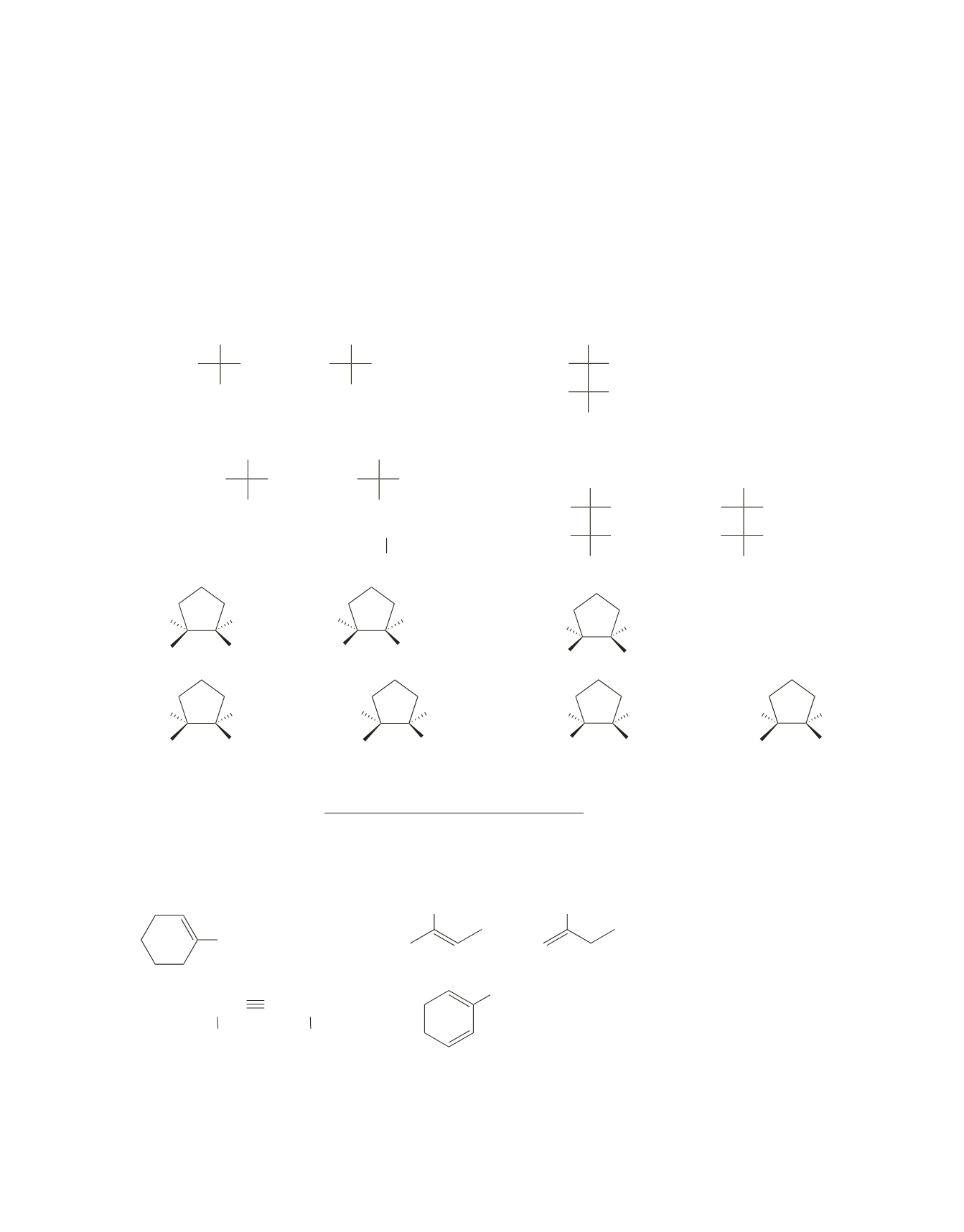
872 Answers
Copyright © 2017 Pearson Education, Inc.
7.
a.
The addition of
Br
2
to 1-butene to form 1,2-dibromobutane is a concerted reaction.
F
b.
The reaction of 1-butene with HCl forms 1-chlorobutane as the major product.
F
c.
The reaction of HBr with 3-methylcyclohexene is more highly regioselective
than is the reaction of HBr with 1-methylcyclohexene.
F
d.
The reaction of an alkene with a carboxylic acid forms an epoxide.
F
e.
A catalyst increases the equilibrium constant of a reaction.
F
f.
The addition of HBr to 3-methyl-2-pentene is a stereospecific reaction.
F
g.
The addition of HBr to 3-methyl-2-pentene is a stereoselective reaction.
F
h.
The addition of HBr to 3-methyl-2-pentene is a regioselective reaction.
T
8.
a.
Cl
H
CH
3
CH
3
CH
2
Cl
H
CH
2
CH
3
CH
3
+
c.
CH
2
CH
3
H Br
CH
2
CH
3
H Br
b.
Br
H
CH
3
CH
3
CH
2
CH
2
Br
H
CH
2
CH
2
CH
3
CH
3
+
+
CH
3
CH
2
CHCH
2
CH
3
Br
d.
CH
2
CH
3
H Br
CH
2
CH
2
CH
3
H Br
CH
2
CH
3
Br
H
CH
2
CH
2
CH
3
Br
H
+
9.
a.
Br
CH
3
Br
H
3
C
+
H
3
C
Br
CH
3
Br
c.
H
3
C
CH
3
H
H
b.
Br
CH
2
CH
3
Br
H
3
C
+
CH
3
CH
2
Br
CH
3
Br
d.
H
3
C
CH
2
CH
3
H
H
+
CH
3
CH
2
CH
3
H
H
Answers to Chapter 7 Practice Test
1.
a. 1.
R
2
BH
>
THF
2.
HO
-
, H
2
O
2
, H
2
O
b.
H
2
>
Lindlar
catalyst
2.
a.
OH
b.
OH
OH
and
3.
CH
3
CH
2
CHC CCH
2
CHCH
3
CH
3
CH
3
a.
b.
CH
3
4.
a.
A terminal alkyne is more stable than an internal alkyne.
F
b.
Propyne is more reactive than propene toward reaction with HBr.
F
c.
1-Butyne is more acidic than 1-butene.
T
















