
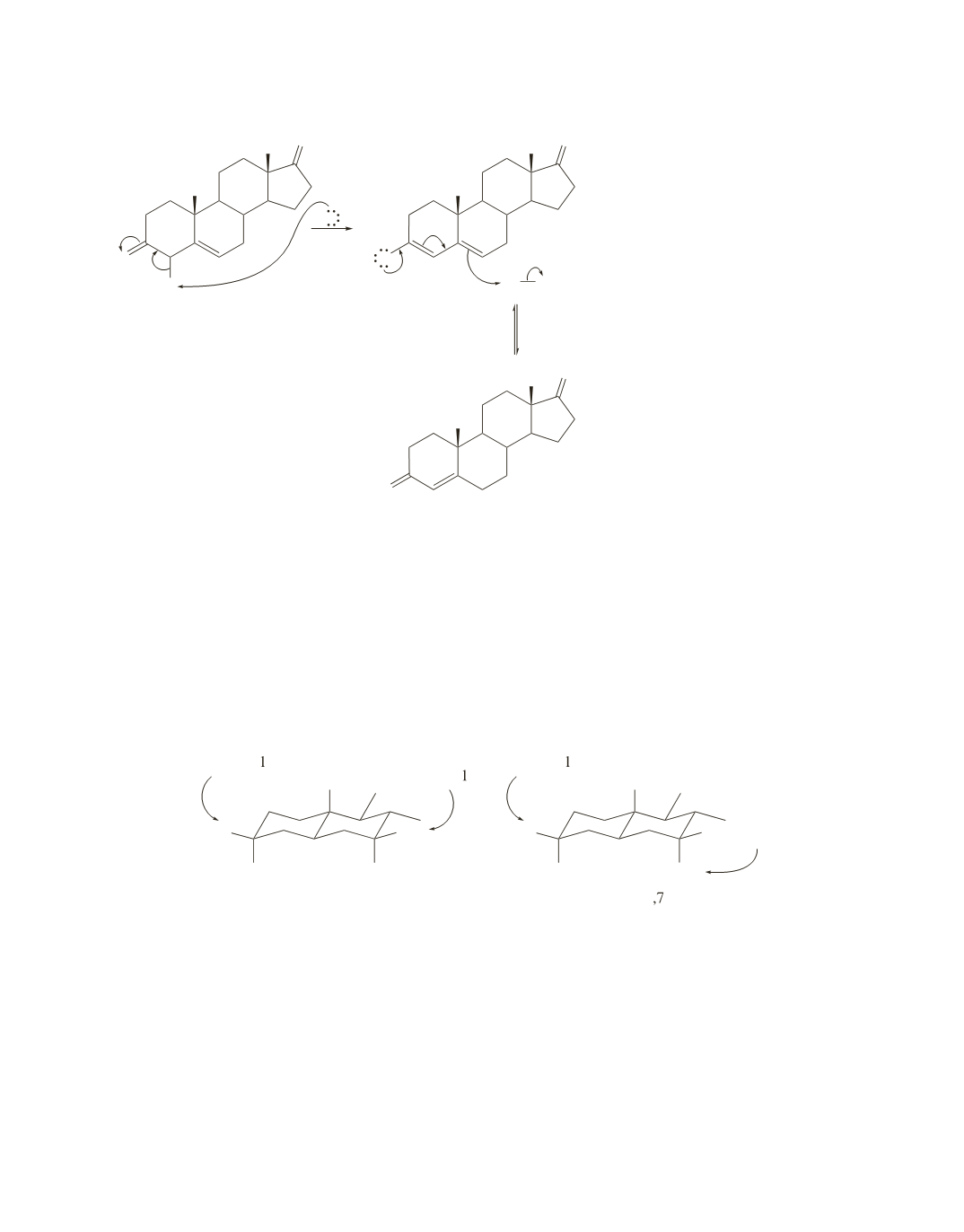
804 Chapter 25
Copyright © 2017 Pearson Education, Inc.
43.
H
O
O
CH
3
CH
3
HO
−
O
O
CH
3
CH
3
−
H OH
O
O
CH
3
CH
3
+
HO
−
44.
The OH groups will react only if they are in equatorial positions, because introduction of bulky axial
substituents would decrease the stability of the molecule.
In the case of 5
a
-cholestane-3
b
,7
b
-diol, the two OH groups are on the same side of the ring system as the
angular methyl group, which means that they are in equatorial positions. Both OH groups react with ethyl
chloroformate.
In the case of 5
a
-cholestane-3
b
,7
a
-diol, only one of the OH groups is on the same side of the ring system
as the angular methyl group. The other is on the opposite side of the ring, which means that it is in an axial
position. Only the OH group that is in the equatorial position reacts with ethyl chloroformate.
OH
H
HO
H
CH
3
equatoria
equatoria
5
A
-cholestane-3
B
,7
B
-diol
H
H
HO
OH
CH
3
equatoria
5
A
-cholestane-3
B A
-diol
axial
45.
The mechanism for the conversion of isopentenyl pyrophosphate to geranyl pyrophospate to farnesyl
pyrophosphate is shown in the text on pages 1143 and 1145. Now we need to show how farnesyl pyrophos-
phate is converted to eudesmol.
















