
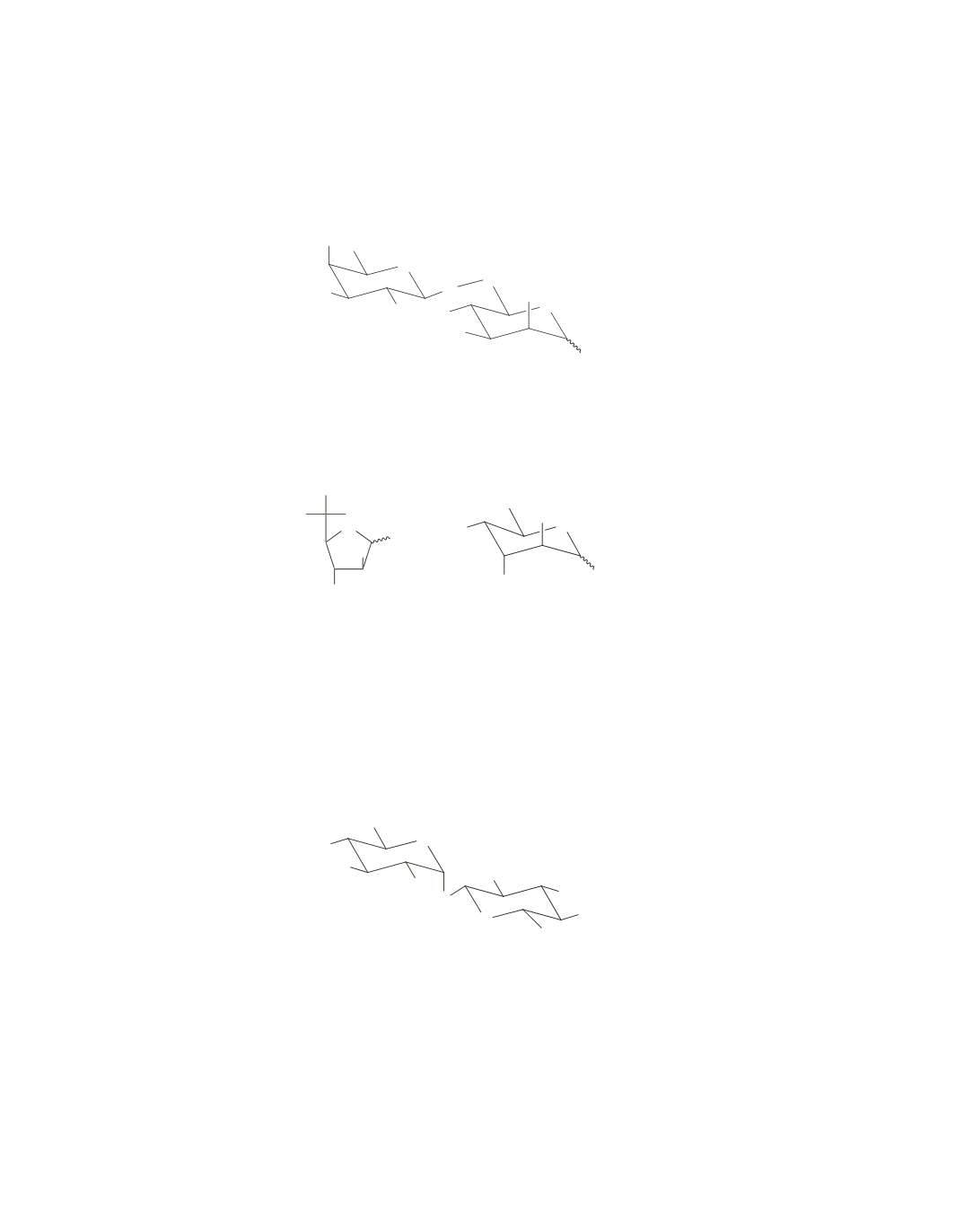
698 Chapter 20
Copyright © 2017 Pearson Education, Inc.
68.
Silver oxide increases the leaving tendency of the iodide ion from methyl iodide, thereby allowing the
nucleophilic substitution reaction to take place with the poorly nucleophilic alcohol groups. Because man-
nose is missing the methyl substituent on the oxygen at C-6, the disaccharide must be formed using the C-6
OH group of mannose and the anomeric carbon of galactose.
O
OH
CH
2
OH
O
OH
O
HO
HO
HO
CH
2
HO
HO
D
-galactose
D
-mannose
69.
d-Altrose most likely exists as a furanose because:
(1)
the furanose is particularly stable since all the large substituents are trans to each other, and
(2)
the pyranose has two of its OH groups in the unstable axial position.
O
OH
HO
HO
OH H
CH
2
OH
D
-altrofuranose
D
-altropyranose
O
HO
OH
OH
OH
CH
2
OH
70.
From its molecular formula and the fact that only glucose is formed when trehalose is hydrolyzed, we
know that it is a disaccharide. Trehalose can be a nonreducing sugar only if the anomeric carbon of one
glucose is connected to the anomeric carbon of the other glucose. Because it can be hydrolyzed by maltase,
its glycosidic linkage must have the same geometry as an
a
-1,4 -glycosidic linkage. Therefore, the OH
group attached to the anomeric carbon of the glucose on the left must be axial, and the OH group attached
to the anomeric carbon of the glucose on the right must be equatorial. (Notice that the glucose on the right
is drawn upside down and is reversed horizontally.)
O
OH
OH
HO
O
HO
HO
OH
O
CH
2
OH
CH
2
OH
















